The two products are not interchangeable “versions of the same thing”: they differ fundamentally in molecular class (peptide vs non‑peptide), oral absorption strategy, dosing constraints, pharmacokinetics, and drug–drug interaction profile. The practical headline for clinicians and patients is that Wegovy pill has stricter administration rules (fasting + limited water + 30‑minute delay before food/other meds), whereas Foundayo can be taken once daily with or without food—a real-world adherence advantage that regulators and market observers have explicitly highlighted.
Efficacy comparisons require discipline. There are no head‑to‑head obesity trials of Wegovy pill vs Foundayo, and the pivotal programs differed in size, duration, and populations. Still, across pivotal placebo‑controlled obesity trials reported in FDA labeling, Wegovy pill (Week 64) produced −13.6% mean body-weight change vs −2.4% with placebo, while Foundayo (Week 72; highest approved dose) produced −11.1% vs −2.1% with placebo. These are both “double‑digit” results, but they should be interpreted as cross‑trial, non‑comparative.
Access is shifting quickly. As of March 2026, Centers for Medicare & Medicaid Services has an explicit policy pathway to support Wegovy (injection and tablets) access for weight loss for eligible beneficiaries through the Medicare GLP‑1 Bridge demonstration beginning July 1, 2026 (separate from Part D), and longer-term via BALANCE starting in 2027. Foundayo is newly approved for obesity and is not listed among CMS Bridge-covered products in the March 2026 CMS FAQ language, so Medicare obesity coverage for Foundayo remains uncertain and should be rechecked as CMS updates program eligibility.
Comparison table
| Attribute | Wegovy pill (oral semaglutide) | Foundayo (orforglipron) |
|---|---|---|
| Molecular class | Peptide GLP‑1 analog; produced by yeast fermentation; albumin binding via fatty-acid modification supports long duration. | Non‑peptide GLP‑1 receptor agonist (orforglipron calcium salt; small-molecule chemical structure). |
| Oral formulation strategy | Co-formulated with SNAC (salcaprozate sodium) absorption enhancer; absorption occurs predominantly in the stomach. | No absorption enhancer required; no clinically relevant food effect reported in labeling. |
| Administration constraints | Empty stomach in the morning with ≤4 oz water, then wait ≥30 minutes before food, beverages, or other oral meds. | Once daily, with or without food; swallow whole. |
| Approved indication focus | Chronic weight management; also approved for CV risk reduction in adults with known heart disease and overweight/obesity in Wegovy labeling. | Chronic weight management in adults with obesity or overweight with ≥1 weight-related comorbidity; use with diet + activity. |
| Pivotal obesity trial (label) | Study 7: 64 weeks, adults with obesity/overweight + comorbidity; T2D excluded. | Trial 1: 72 weeks, adults with obesity/overweight + comorbidity; T2D excluded. |
| Mean weight loss (pivotal obesity trial) | −13.6% (Wegovy pill) vs −2.4% placebo at Week 64. | −11.1% (Foundayo 17.2 mg) vs −2.1% placebo at Week 72. |
| Glycemic outcomes in pivotal obesity trial | Not a central labeled endpoint for the tablet obesity pivotal trial; trial excluded T2D. | Includes a separate 72‑week trial in T2D with HbA1c reductions up to −1.7% (17.2 mg) vs −0.4% placebo. |
| Pharmacokinetics | Oral semaglutide bioavailability ~1–2%; Cmax reached ~1 hour post-dose; semaglutide half-life ~1 week. | Absolute bioavailability 77% (0.8 mg); Cmax 4–8 hours post-dose; half-life ~29–49 hours. |
| Drug–drug interactions | Can affect absorption of oral meds; levothyroxine exposure increased 33% in a tablet interaction study (monitor). | Clinically relevant interactions: dose limits with strong CYP3A4 inhibitors, avoid strong inducers; simvastatin acid ↑2–2.5×; guidance for oral contraceptives due to gastric emptying delay. |
| Storage | Tablets: controlled room temp; keep in original bottle; protect from moisture. | Room temp; light sensitive—protect from light (original bottle/carton). |
| Pricing signals (US) | Novo Nordisk website lists self‑pay program pricing such as $149/month for certain early tablet strengths (program terms apply). | Market reporting indicates self‑pay launch pricing starting near $149/month via direct channel, with higher-dose pricing tiers; terms vary and should be verified at time of prescribing. |
| Label boxed warning & key contraindication | Boxed warning: thyroid C‑cell tumors; contraindicated in personal/family history of MTC or MEN2. | Boxed warning: thyroid C‑cell tumors; same contraindication concept (MTC/MEN2) plus hypersensitivity contraindication. |
Drug profiles and formulation science
Wegovy pill uses semaglutide, a peptide GLP‑1 analog engineered for prolonged action (notably via albumin binding) and manufactured with a peptide backbone produced by yeast fermentation. The FDA labeling explicitly describes these structural features and their role in protraction.
The central challenge for oral semaglutide has always been oral bioavailability—peptides are typically degraded in the GI tract and do not readily cross membranes. Wegovy tablets address this with salcaprozate sodium (SNAC), an absorption enhancer co-formulated in the tablet; labeling states absorption occurs predominantly in the stomach and that absolute bioavailability is only ~1–2% even with this approach. The clinical consequence is that administration conditions materially influence exposure (water volume and post-dose fasting duration).
Foundayo uses orforglipron calcium, described in labeling by molecular formula and a defined chemical structure, consistent with a non‑peptide small molecule that directly activates the human GLP‑1 receptor. Unlike oral semaglutide, Foundayo is not dependent on an absorption enhancer and shows no clinically relevant food effect in labeling—one reason it can be dosed without fasting constraints.
From a scalability and distribution standpoint, both products are solid oral dosage forms stored at controlled room temperature, but each has different handling sensitivities (Wegovy tablets: moisture; Foundayo: light). These details matter operationally for pharmacies and for patient storage behavior.
Regulatory status and market availability
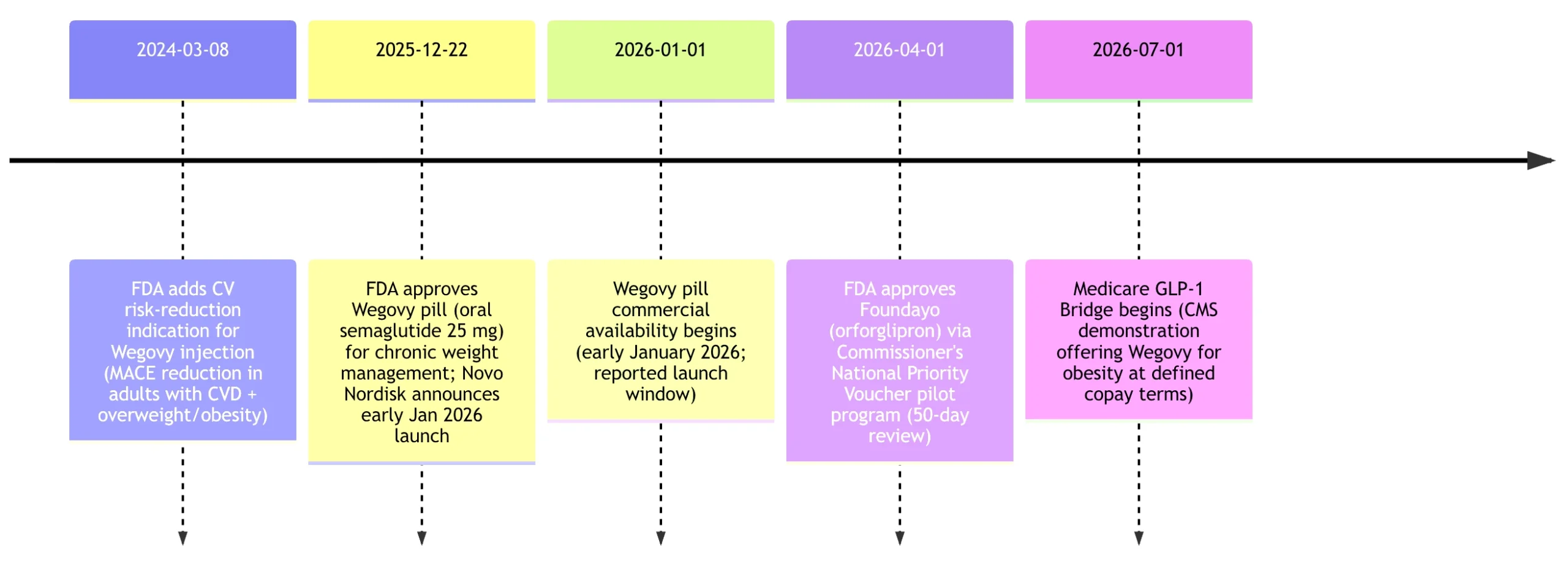
In the United States, Wegovy pill entered the market first: Novo Nordisk announced FDA approval December 22, 2025 and an expected US launch in early January 2026.
Foundayo was approved by the FDA on April 1, 2026 and is historically notable not just as a new weight-loss pill but as an accelerated regulatory experiment. The FDA states the decision was issued 50 days after filing, 294 days before the application’s stated PDUFA date (January 20, 2027), and represents the fastest NME approval since 2002. The agency also characterizes this as the first new molecular entity approved under the voucher program and the fifth approval overall under the Commissioner’s National Priority Voucher pilot program.
Outside the US, the situation is less mature for the oral products. Wegovy (semaglutide) is established in Europe in injectable forms (EMA EPAR exists for Wegovy), and Novo Nordisk has publicly stated it submitted oral semaglutide 25 mg for obesity to the EMA and other regulators in the second half of 2025; an EMA CHMP agenda item in February 2026 indicates ongoing committee activity around Wegovy/semaglutide.
In the United Kingdom, recent regulatory news has focused on injectable semaglutide dose expansions (e.g., the UK regulator’s announcement about higher-dose Wegovy injection), rather than an approved “Wegovy pill.”
For Foundayo, the FDA approval is US-specific as of April 1, 2026, while Lilly has described a multi-country regulatory strategy (submissions and plans across many jurisdictions), but major non‑US approvals were not identified in the primary sources reviewed here.
The March 2024 milestone reflects the FDA press announcement about the cardiovascular indication for Wegovy injection; the July 2026 milestone reflects CMS’s demonstration timeline and eligibility description.
Key milestones for oral semaglutide and orforglipron (US-centric)
Clinical trial evidence and outcomes
The evidence bases differ in shape. Wegovy pill’s pivotal obesity evidence in labeling is built around a smaller, 64‑week randomized placebo-controlled trial (Study 7) in adults with obesity/overweight and comorbidity, excluding type 2 diabetes, while Foundayo’s pivotal obesity evidence in labeling relies on two large 72‑week placebo‑controlled trials, one without diabetes and one with type 2 diabetes.
Wegovy pill pivotal obesity trial in labeling
In the Wegovy label, the tablet obesity study enrolled adults with obesity or overweight plus at least one weight-related comorbidity, excluded type 2 diabetes, and followed a titration to 25 mg daily with a total 64-week treatment period (12-week escalation + 52-week maintenance). Baseline mean body weight was ~106 kg and mean BMI ~37.5 kg/m² in the tablet trial population described in the label.
At Week 64, the label reports an LSMean −13.6% body-weight change for Wegovy pill versus −2.4% for placebo (difference about −11.2%), with 76.3% achieving ≥5% weight loss versus 31.3% with placebo.
Foundayo pivotal obesity trials in labeling
Foundayo’s label describes Trial 1 (without diabetes) as a 72‑week randomized placebo-controlled study enrolling 3,127 adults with obesity or overweight plus at least one comorbidity (including dyslipidemia, hypertension, obstructive sleep apnea, or cardiovascular disease) and excluding type 2 diabetes.
At Week 72 in Trial 1, mean percent change in body weight was −11.1% for Foundayo 17.2 mg once daily versus −2.1% for placebo (difference −9.0%), with 71.5% achieving ≥5% weight loss versus 26.8% on placebo.
Trial 2 enrolled 1,613 adults with BMI ≥27 kg/m² and type 2 diabetes and assessed both weight and glycemic outcomes. At Week 72, the 17.2 mg group showed −9.6% weight change vs −2.5% with placebo, and HbA1c changes to −1.7% vs −0.4% with placebo (difference −1.2% for the 17.2 mg arm vs placebo).
Interpreting comparative efficacy
The figure displayed above visualizes mean weight change in the pivotal obesity trials as reported in FDA labeling. It is useful for orientation, but it should not be read as a head-to-head claim because trial durations and designs differ (Week 64 vs Week 72; different sample sizes and demographics; potentially different behavior programs).
A separate “head‑to‑head” evidence stream exists for type 2 diabetes, where Lilly has reported results from an orforglipron vs oral semaglutide trial program; however, those comparisons used oral semaglutide diabetes doses (e.g., 7 mg/14 mg) rather than the 25 mg obesity-dose Wegovy pill, and therefore should be treated as indirect evidence about the oral-class tradeoffs rather than a direct answer to “Wegovy pill vs Foundayo for obesity.”
Dosing, PK/PD, and real-world adherence
Dose escalation and administration
Wegovy pill follows a fixed titration to reach 25 mg daily: 1.5 mg daily days 1–30, 4 mg days 31–60, 9 mg days 61–90, then 25 mg daily maintenance thereafter.
Critically, Wegovy pill must be taken in a narrow administration window: on an empty stomach in the morning with up to 4 ounces of water, and the patient must then wait at least 30 minutes before eating, drinking, or taking other oral medications. The label also presents pharmacokinetic evidence that water volume and post-dose fasting duration can materially affect exposure (AUC/Cmax).
Foundayo’s labeled escalation is more flexible in its upper range: start 0.8 mg daily, then after ≥30 days move to 2.5 mg, after ≥30 days to 5.5 mg, and then optional increases (after ≥30 days at each level) to 9 mg, 14.5 mg, or 17.2 mg based on response and tolerability. It is taken once daily with or without food.
Pharmacokinetics and practical implications
Wegovy pill’s core PK constraint is low and variable oral bioavailability. The label estimates oral semaglutide absolute bioavailability at ~1–2%, with peak concentration around 1 hour post-dose, and a long semaglutide half-life of ~1 week, implying drug persistence for weeks after discontinuation.
Foundayo shows “small‑molecule‑like” oral PK: labeling reports absolute bioavailability 77% (at a 0.8 mg dose), peak concentration 4–8 hours post-dose, and a half-life of ~29–49 hours.
From a real-world adherence perspective, regulators and market reporting have highlighted that Foundayo’s lack of fasting/water restrictions may help adoption among patients who struggle with strict morning routines, while Wegovy pill’s regimen complexity is a known friction point rooted in oral semaglutide’s absorption biology.
Safety signals, contraindications, and monitoring
Both products carry class-consistent safety issues typical for GLP‑1 receptor agonist activity. Each label includes a boxed warning for thyroid C‑cell tumors and contraindications for personal/family history of medullary thyroid carcinoma (MTC) or MEN2.
Beyond the boxed warning, the FDA’s press summary for Foundayo lists labeled warnings/precautions including pancreatitis, severe GI reactions, acute kidney injury due to volume depletion, hypoglycemia, hypersensitivity, diabetic retinopathy complications (in T2D), gallbladder disease, and pulmonary aspiration with anesthesia/deep sedation.
Wegovy labeling likewise lists multiple warnings and precautions and explicitly notes rare postmarketing pulmonary aspiration reports in patients on GLP‑1 agents undergoing anesthesia/deep sedation, reflecting a broader anesthesia-safety discussion around delayed gastric emptying.
Discontinuation due to adverse reactions in the pivotal tablet obesity trial was 6.9% for Wegovy pill vs 5.9% for placebo; Foundayo labeling reports 8% discontinuations due to adverse reactions vs 3% placebo across both pivotal trials, with GI adverse reactions a major driver in both programs.
A differentiator is drug–drug interaction complexity. Wegovy tablet labeling warns about delayed gastric emptying effects and documents increased levothyroxine exposure (monitoring recommended). Foundayo labeling adds a more “small‑molecule‑like” interaction profile—dose modifications for CYP3A4 modulators and a substantial increase in simvastatin acid exposure (dose limits), plus guidance to use non-oral or barrier contraception around dose changes due to potential effects on oral medication absorption.
Pricing, insurance coverage, and access
Pricing and coverage for obesity pharmacotherapy remain unusually dynamic, and sources often mix list price, net price, and patient out‑of‑pocket programs. The most reliable approach is to separate: (1) manufacturer cash-pay programs, (2) commercial insurance copay support, and (3) public payer eligibility.
For Wegovy pill, the manufacturer’s US consumer site describes programmatic cash-pay pricing (example: $149/month for certain introductory tablet strengths under stated terms and time windows) and also markets commercial savings (e.g., “pay as little as $25,” eligibility dependent). These offers are explicitly time-limited and modifiable by the manufacturer.
For Foundayo, market reporting around launch describes a cash-pay entry price around $149/month via a direct channel, with higher tiers for higher doses and separate copay scenarios for insured patients; because these are programmatic and early-launch numbers, they should be verified at the time of prescribing.
Public coverage policy is changing as of 2026. CMS’s March 2026 “Medicare GLP‑1 Bridge” policy describes a nationwide demonstration operating July 1–December 31, 2026 that provides eligible beneficiaries early access to certain GLP‑1 drugs for weight loss outside the normal Part D payment flow; at the time of the CMS FAQ text, eligible products for the Bridge are Wegovy (injection and tablets) and Zepbound, with a stated beneficiary cost-sharing structure and additional operational details (including a central processor administered by Humana). Foundayo is not identified as an eligible product in the CMS Bridge FAQ text provided.
Independent policy analysis from KFF frames this as a two-step expansion: the 2026 Bridge followed by the BALANCE Model beginning in 2027, while also emphasizing that Medicaid coverage for obesity drugs remains optional by state (with limited state coverage as of early 2026).
Because payer policy is moving, the most defensible “access” summary for April 2026 is:
Wegovy pill: FDA-approved and commercially launched; notably positioned for Medicare access via CV indication (Part D) plus explicit Bridge eligibility for obesity beginning July 2026 (per CMS).
Foundayo: FDA-approved and launching via commercial channels; public payer access for obesity depends on evolving Medicare/Medicaid rules and program eligibility updates not yet reflected in the March 2026 CMS Bridge eligibility statement.